A full list of our publications is available at PubMed.
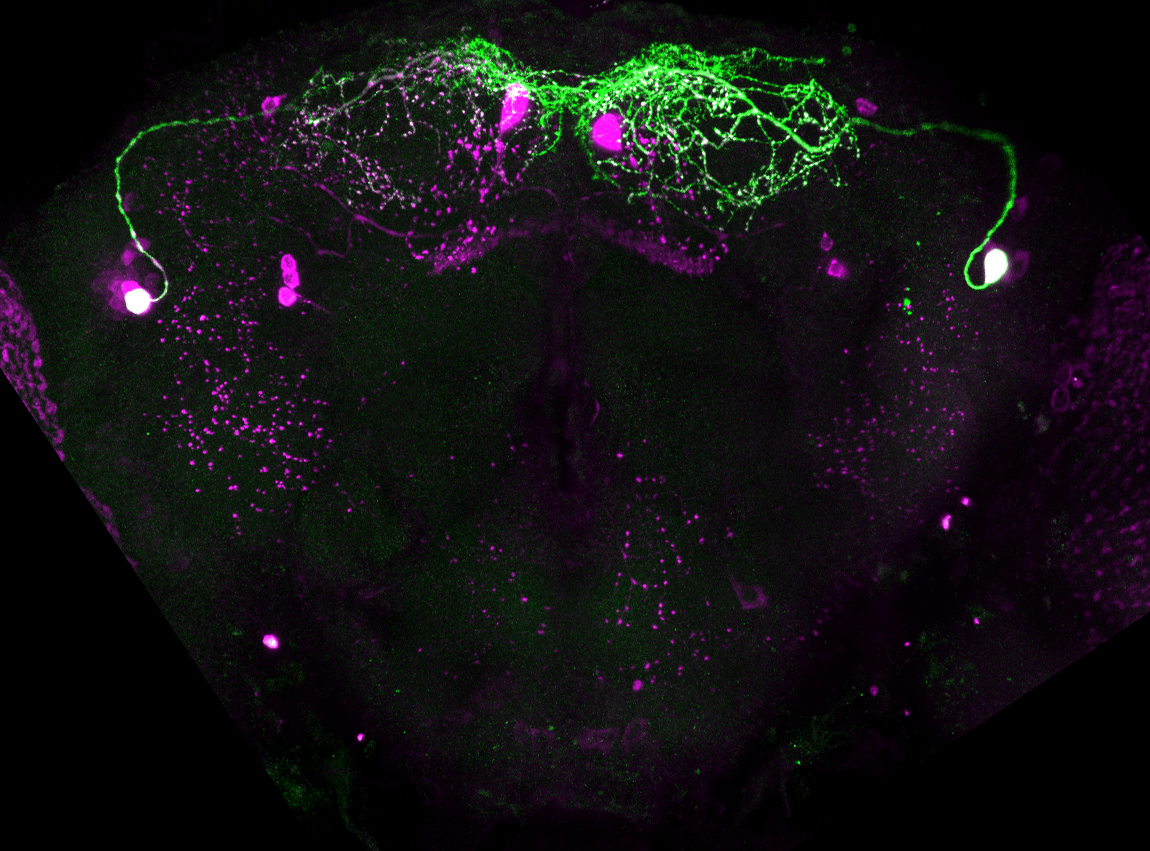
A single NPFR neuropeptide F receptor neuron that regulates thirst behaviors in Drosophila
Thirst is a strongly motivated internal state that is represented in central brain circuits that are only partially understood. Water seeking is a discrete step of the thirst behavioral sequence that is amenable to uncovering the mechanisms for motivational properties such as goal-oriented behavior, value encoding, and behavioral competition. In Drosophila water seeking is regulated by the NPY-like neuropeptide NPF, however the circuitry for NPF-dependent water seeking is unknown. To uncover the downstream circuitry, we identified the NPF receptor NPFR and the neurons it is expressed in as being acutely critical for thirsty water seeking in males. Refinement of the NPFR pattern uncovered a role for a single neuron, the L1-l, in promoting thirsty water seeking. The L1-l neuron increases its activity in thirsty flies and is involved in the regulation of dopaminergic neurons in long-term memory formation. Thus, NPFR and its ligand NPF, already known for its role in feeding behavior, are also important for a second ingestive behavior.
PMID: 41062275 Download: PDF
Memory-like states created by the first ethanol experience are encoded into the Drosophila mushroom body learning and memory circuitry in an ethanol-specific manner
A first ethanol exposure creates three memory-like states in Drosophila. Ethanol memory-like states appear genetically and behaviorally paralleled to the canonical learning and memory traces anesthesia-sensitive, anesthesia-resistant, and long-term memory ASM, ARM, and LTM. It is unknown if these ethanol memory-like states are also encoded by the canonical learning and memory circuitry that is centered on the mushroom bodies. We show that the three ethanol memory-like states, anesthesia-sensitive tolerance (AST) and anesthesia resistant tolerance (ART) created by ethanol sedation to a moderately high ethanol exposure, and chronic tolerance created by a longer low concentration ethanol exposure, each engage the mushroom body circuitry differently. Moreover, critical encoding steps for ethanol memory-like states reside outside the mushroom body circuitry, and within the mushroom body circuitry they are markedly distinct from classical memory traces. Thus, the first ethanol exposure creates distinct memory-like states in ethanol-specific circuits and impacts the function of learning and memory circuitry in ways that might influence the formation and retention of other memories.
PMID: 39899623 Download: PDF
OTUD6 deubiquitination of RPS7/eS7 on the free 40 S ribosome regulates global protein translation and stress
Ribosomes are regulated by evolutionarily conserved ubiquitination/deubiquitination events. We uncover the role of the deubiquitinase OTUD6 in regulating global protein translation through deubiquitination of the RPS7/eS7 subunit on the free 40 S ribosome in vivo in Drosophila. Coimmunoprecipitation and enrichment of monoubiquitinated proteins from catalytically inactive OTUD6 flies reveal RPS7 as the ribosomal substrate. The 40 S protein RACK1 and E3 ligases CNOT4 and RNF10 function upstream of OTUD6 to regulate alkylation stress. OTUD6 interacts with RPS7 specifically on the free 40 S, and not on 43 S/48 S initiation complexes or the translating ribosome. Global protein translation levels are bidirectionally regulated by OTUD6 protein abundance. OTUD6 protein abundance is physiologically regulated in aging and in response to translational and alkylation stress. Thus, OTUD6 may promote translation initiation, the rate limiting step in protein translation, by titering the amount of 40 S ribosome that recycles.
PMID: 39127721 Download: PDF
Drosophila learning and memory centers and the actions of drugs of abuse
Drug addiction and the circuitry for learning and memory are intimately intertwined. Drugs of abuse create strong, inappropriate, and lasting memories that contribute to many of their destructive properties, such as continued use despite negative consequences and exceptionally high rates of relapse. Studies in Drosophila melanogaster are helping us understand how drugs of abuse, especially alcohol, create memories at the level of individual neurons and in the circuits where they function. Drosophila is a premier organism for identifying the mechanisms of learning and memory. Drosophila also respond to drugs of abuse in ways that remarkably parallel humans and rodent models. An emerging consensus is that, for alcohol, the mushroom bodies participate in the circuits that control acute drug sensitivity, not explicitly associative forms of plasticity such as tolerance, and classical associative memories of their rewarding and aversive properties. Moreover, it is becoming clear that drugs of abuse use the mushroom body circuitry differently from other behaviors, potentially providing a basis for their addictive properties.
PMID: 38862166 Download: PDF
Alcohol sensitivity and tolerance encoding in sleep regulatory circadian neurons in Drosophila
Alcohol tolerance is a simple form of behavioural and neural plasticity that occurs with the first drink. Neural plasticity in tolerance is likely a substrate for longer term adaptations that can lead to alcohol use disorder. Drosophila develop tolerance with characteristics similar to vertebrates, and it is a useful model for determining the molecular and circuit encoding mechanisms in detail. Rapid tolerance, measured after the first alcohol exposure is completely metabolized, is localized to specific brain regions that are not interconnected in an obvious way. We used a forward neuroanatomical screen to identify three new neural sites for rapid tolerance encoding. One of these was composed of two groups of neurons, the DN1a and DN1p glutamatergic neurons, that are part of the Drosophila circadian clock. We localized rapid tolerance to the two DN1a neurons that regulate arousal by light at night, temperature-dependent sleep timing, and night-time sleep. Two clock neurons that regulate evening activity, LNd6 and the 5th LNv, are postsynaptic to the DN1as, and they promote rapid tolerance via the metabotropic glutamate receptor. Thus, rapid tolerance to alcohol overlaps with sleep regulatory neural circuitry, suggesting a mechanistic link.
PMID: 37500483 Download: PDF
Rapid and chronic ethanol tolerance are composed of distinct memory-like states in Drosophila
Ethanol tolerance is the first type of behavioral plasticity and neural plasticity that is induced by ethanol intake, and yet its molecular and circuit bases remain largely unexplored. Here, we characterize three distinct forms of ethanol tolerance in male Drosophila: rapid, chronic, and repeated. Rapid tolerance is composed of two short-lived memory-like states, one that is labile and one that is consolidated. Chronic tolerance, induced by continuous exposure, lasts for two days, induces ethanol preference, and hinders the development of rapid tolerance through the activity of histone deacetylases (HDACs). Unlike rapid tolerance, chronic tolerance is independent of the immediate early gene Hr38/Nr4a. Chronic tolerance is suppressed by the Sirtuin HDAC Sirt1, whereas rapid tolerance is enhanced by Sirt1. Moreover, rapid and chronic tolerance map to anatomically distinct regions of the mushroom body learning and memory centers. Chronic tolerance, like long term memory, is dependent on new protein synthesis and it induces the kayak/c-fos immediate early gene, but it depends on CREB signaling outside the mushroom bodies, and it does not require the Radish GTPase. Thus, chronic ethanol exposure creates an ethanol-specific memory-like state that is molecularly and anatomically different from other forms of ethanol tolerance.
PMID: 36750369 Download: PDF
Thirst interneurons that promote water seeking and limit feeding behavior in Drosophila
Thirst is a motivational state that drives behaviors to obtain water for fluid homeostasis. We identified two types of central brain interneurons that regulate thirsty water seeking in Drosophila, that we term the Janu neurons. Janu-GABA, a local interneuron in the subesophageal zone, is activated by water deprivation and is specific to thirsty seeking. Janu-AstA projects from the subesophageal zone to the superior medial protocerebrum, a higher order processing area. Janu-AstA signals with the neuropeptide Allatostatin A to promote water seeking and to inhibit feeding behavior. NPF (Drosophila NPY) neurons are postsynaptic to Janu-AstA for water seeking and feeding through the AstA-R2 galanin-like receptor. NPF neurons use NPF to regulate thirst and hunger behaviors. Flies choose Janu neuron activation, suggesting that thirsty seeking up a humidity gradient is rewarding. These findings identify novel central brain circuit elements that coordinate internal state drives to selectively control motivated seeking behavior.
PMID: 34018925 Download: PDF
Mef2 induction of the immediate early gene Hr38/Nr4a is terminated by Sirt1 to promote ethanol tolerance
Drug naïve animals given a single dose of ethanol show changed responses to subsequent doses, including the development of ethanol tolerance and ethanol preference. These simple forms of behavioral plasticity are due in part to changes in gene expression and neuronal properties. Surprisingly little is known about how ethanol initiates changes in gene expression or what the changes do. Here we demonstrate a role in ethanol plasticity for Hr38, the sole Drosophila homolog of the mammalian Nr4a1/2/3 class of immediate early response transcription factors. Acute ethanol exposure induces transient expression of Hr38 and other immediate early neuronal activity genes. Ethanol activates the Mef2 transcriptional activator to induce Hr38, and the Sirt1 histone/protein deacetylase is required to terminate Hr38 induction. Loss of Hr38 decreases ethanol tolerance and causes precocious but short-lasting ethanol preference. Similarly, reduced Mef2 activity in all neurons or specifically in the mushroom body α/β neurons decreases ethanol tolerance; Sirt1 promotes ethanol tolerance in these same neurons. Genetically decreasing Hr38 expression levels in Sirt1 null mutants restores ethanol tolerance, demonstrating that both induction and termination of Hr38 expression are important for behavioral plasticity to proceed. These data demonstrate that Hr38 functions as an immediate early transcription factor that promotes ethanol behavioral plasticity.
PMID: 29726098 Download: PDF
Satiation State-dependent Dopaminergic Control of Foraging in Drosophila
Hunger evokes stereotypic behaviors that favor the discovery of nutrients. The neural pathways that coordinate internal and external cues to motivate foraging behaviors are only partly known. Drosophila that are food deprived increase locomotor activity, are more efficient in locating a discrete source of nutrition, and are willing to overcome adversity to obtain food. We developed a simple open field assay that allows flies to freely perform multiple steps of the foraging sequence, and we show that two distinct dopaminergic neural circuits regulate measures of foraging behaviors. One group, the PAM neurons, functions in food deprived flies while the other functions in well fed flies, and both promote foraging. These satiation state-dependent circuits converge on dopamine D1 receptor-expressing Kenyon cells of the mushroom body, where neural activity promotes foraging independent of satiation state. These findings provide evidence for active foraging in well-fed flies that is separable from hunger-driven foraging.
PMID: 29636522 Download: PDF
Perineurial Barrier Glia Physically Respond to Alcohol in an Akap200-Dependent Manner to Promote Tolerance
Ethanol is the most common drug of abuse. It exerts its behavioral effects by acting on widespread neural circuits; however, its impact on glial cells is less understood. We show that Drosophila perineurial glia are critical for ethanol tolerance, a simple form of behavioral plasticity. The perineurial glia form the continuous outer cellular layer of the blood-brain barrier and are the interface between the brain and the circulation. Ethanol tolerance development requires the A kinase anchoring protein Akap200 specifically in perineurial glia. Akap200 tightly coordinates protein kinase A, actin, and calcium signaling at the membrane to control tolerance. Furthermore, ethanol causes a structural remodeling of the actin cytoskeleton and perineurial membrane topology in an Akap200-dependent manner, without disrupting classical barrier functions. Our findings reveal an active molecular signaling process in the cells at the blood-brain interface that permits a form of behavioral plasticity induced by ethanol.
PMID: 29444420 Download: PDF
Sir2/Sirt1 Links Acute Inebriation to Presynaptic Changes and the Development of Alcohol Tolerance, Preference, and Reward
Acute ethanol inebriation causes neuroadaptive changes in behavior that favor increased intake. Ethanol-induced alterations in gene expression, through epigenetic and other means, are likely to change cellular and neural circuit function. Ethanol markedly changes histone acetylation, and the sirtuin Sir2/SIRT1 that deacetylates histones and transcription factors is essential for the rewarding effects of long-term drug use. The molecular transformations leading from short-term to long-term ethanol responses mostly remain to be discovered. We find that Sir2 in the mushroom bodies of the fruit fly Drosophila promotes short-term ethanol-induced behavioral plasticity by allowing changes in the expression of presynaptic molecules. Acute inebriation strongly reduces Sir2 levels and increases histone H3 acetylation in the brain. Flies lacking Sir2 globally, in the adult nervous system, or specifically in the mushroom body α/β-lobes show reduced ethanol sensitivity and tolerance. Sir2-dependent ethanol reward is also localized to the mushroom bodies, and Sir2 mutants prefer ethanol even without a priming ethanol pre-exposure. Transcriptomic analysis reveals that specific presynaptic molecules, including the synaptic vesicle pool regulator Synapsin, depend on Sir2 to be regulated by ethanol. Synapsin is required for ethanol sensitivity and tolerance. We propose that the regulation of Sir2/SIRT1 by acute inebriation forms part of a transcriptional program in mushroom body neurons to alter presynaptic properties and neural responses to favor the development of ethanol tolerance, preference, and reward.
PMID: 27170122 Download: PDF
Shared neurocircuitry underlying feeding and drugs of abuse in Drosophila
The neural circuitry and molecules that control the rewarding properties of food and drugs of abuse appear to partially overlap in the mammalian brain. This has raised questions about the extent of the overlap and the precise role of specific circuit elements in reward and in other behaviors associated with feeding regulation and drug responses. The much simpler brain of invertebrates including the fruit fly Drosophila, offers an opportunity to make high-resolution maps of the circuits and molecules that govern behavior. Recent progress in Drosophila has revealed not only some common substrates for the actions of drugs of abuse and for the regulation of feeding, but also a remarkable level of conservation with vertebrates for key neuromodulatory transmitters. We speculate that Drosophila may serve as a model for distinguishing the neural mechanisms underlying normal and pathological motivational states that will be applicable to mammals.
PMID: 27013449 Download: PDF
A Pair of Dopamine Neurons Target the D1-Like Dopamine Receptor DopR in the Central Complex to Promote Ethanol-Stimulated Locomotion in Drosophila
Dopamine is a mediator of the stimulant properties of drugs of abuse, including ethanol, in mammals and in the fruit fly Drosophila. The neural substrates for the stimulant actions of ethanol in flies are not known. We show that a subset of dopamine neurons and their targets, through the action of the D1-like dopamine receptor DopR, promote locomotor activation in response to acute ethanol exposure. A bilateral pair of dopaminergic neurons in the fly brain mediates the enhanced locomotor activity induced by ethanol exposure, and promotes locomotion when directly activated. These neurons project to the central complex ellipsoid body, a structure implicated in regulating motor behaviors. Ellipsoid body neurons are required for ethanol-induced locomotor activity and they express DopR. Elimination of DopR blunts the locomotor activating effects of ethanol, and this behavior can be restored by selective expression of DopR in the ellipsoid body. These data tie the activity of defined dopamine neurons to D1-like DopR-expressing neurons to form a neural circuit that governs acute responding to ethanol.
PMID: 20376353 Download: PDF
Ethanol-regulated genes that contribute to ethanol sensitivity and rapid tolerance in Drosophila.
Increased ethanol intake, a major predictor for the development of alcohol use disorders, is facilitated by the development of tolerance to both the aversive and pleasurable effects of the drug. The molecular mechanisms underlying ethanol tolerance development are complex and are not yet well understood. To identify genetic mechanisms that contribute to ethanol tolerance, we examined the time course of gene expression changes elicited by a single sedating dose of ethanol in Drosophila, and completed a behavioral survey of strains harboring mutations in ethanol-regulated genes. Enrichment for genes in metabolism, nucleic acid binding, olfaction, regulation of signal transduction, and stress suggests that these biological processes are coordinately affected by ethanol exposure. We also detected a coordinate up-regulation of genes in the Toll and Imd innate immunity signal transduction pathways. A multi-study comparison revealed a small set of genes showing similar regulation, including increased expression of 3 genes for serine biosynthesis. A survey of Drosophila strains harboring mutations in ethanol-regulated genes for ethanol sensitivity and tolerance phenotypes revealed roles for serine biosynthesis, olfaction, transcriptional regulation, immunity, and metabolism. Flies harboring deletions of the genes encoding the olfactory co-receptor Or83b or the sirtuin Sir2 showed marked changes in the development of ethanol tolerance. Our findings implicate novel roles for these genes in regulating ethanol behavioral responses.
PMID: 19951294 Download: PDF
High-Resolution Analysis of Ethanol-Induced Locomotor Stimulation in Drosophila
Understanding how ethanol influences behavior is key to deciphering the mechanisms of ethanol action and alcoholism. In mammals, low doses of ethanol stimulate locomotion, whereas high doses depress it. The acute stimulant effect of ethanol has been proposed to be a manifestation of its rewarding effects. In Drosophila, ethanol exposure transiently potentiates locomotor activity in a biphasic dose- and time-dependent manner. An initial short-lived peak of activity corresponds to an olfactory response to ethanol. A second, longer-lasting period of increased activity coincides with rising internal ethanol concentrations; these closely parallel concentrations that stimulate locomotion in mammals. High-resolution analysis of the walking pattern of individual flies revealed that locomotion consists of bouts of activity; bout structure can be quantified by bout frequency, bout length, and the time spent walking at high speeds. Ethanol exposure induces both dramatic and dynamic changes in bout structure. Mutants with increased ethanol sensitivity show distinct changes in ethanol-induced locomotor behavior, as well as genotype-specific changes in activity bout structure. Thus, the overall effect of ethanol on locomotor behavior in Drosophila is caused by changes in discrete quantifiable parameters of walking pattern. The effects of ethanol on locomotion are comparable in flies and mammals, suggesting that Drosophila is a suitable model system to study the underlying mechanisms.
PMID: 12486199 Download: PDF
Habituation of an odorant-induced startle response in Drosophila
Habituation is a fundamental form of behavioral plasticity that permits organisms to ignore inconsequential stimuli. Here we describe the habituation of a locomotor response to ethanol and other odorants in Drosophila, measured by an automated high-throughput locomotor tracking system. Flies exhibit an immediate and transient startle response upon exposure to a novel odor. Surgical removal of the antennae, the fly's major olfactory organs, abolishes this startle response. With repeated discrete exposures to ethanol vapor, the startle response habituates. Habituation is reversible by a mechanical stimulus and is not due to the accumulation of ethanol in the organism, nor to non-specific mechanisms. Ablation or inactivation of the mushroom bodies, central brain structures involved in olfactory and courtship conditioning, results in decreased olfactory habituation. In addition, olfactory habituation to ethanol generalizes to odorants that activate separate olfactory receptors. Finally, habituation is impaired in rutabaga, an adenylyl cyclase mutant isolated based on a defect in olfactory associative learning. These data demonstrate that olfactory habituation operates, at least in part, through central mechanisms. This novel model of olfactory habituation in freely moving Drosophila provides a scalable method for studying the molecular and neural bases of this simple and ubiquitous form of learning.
PMID: 15140008 Download: PDF
Invertebrate models of drug abuse.
Susceptibility to drug addiction depends on genetic and environmental factors and their complex interactions. Studies with mammalian models have identified molecular targets, neurochemical systems, and brain regions that mediate some of the addictive properties of abused drugs. Yet, our understanding of how the primary effects of drugs lead to addiction remains incomplete. Recently, researchers have turned to the invertebrate model systems
Drosophila melanogaster and
Caenorhabditis elegans to dissect the mechanisms by which abused drugs modulate behavior. Due to their sophisticated genetics, relatively simple anatomy, and their remarkable molecular similarity to mammals, these invertebrate models should provide useful insights into the mechanisms of drug action. Here we review recent behavioral and genetic studies in flies and worms on the effects of ethanol, cocaine, and nicotine, three of the most widely abused drugs in the world.
PMID: 12486703 Download: PDF